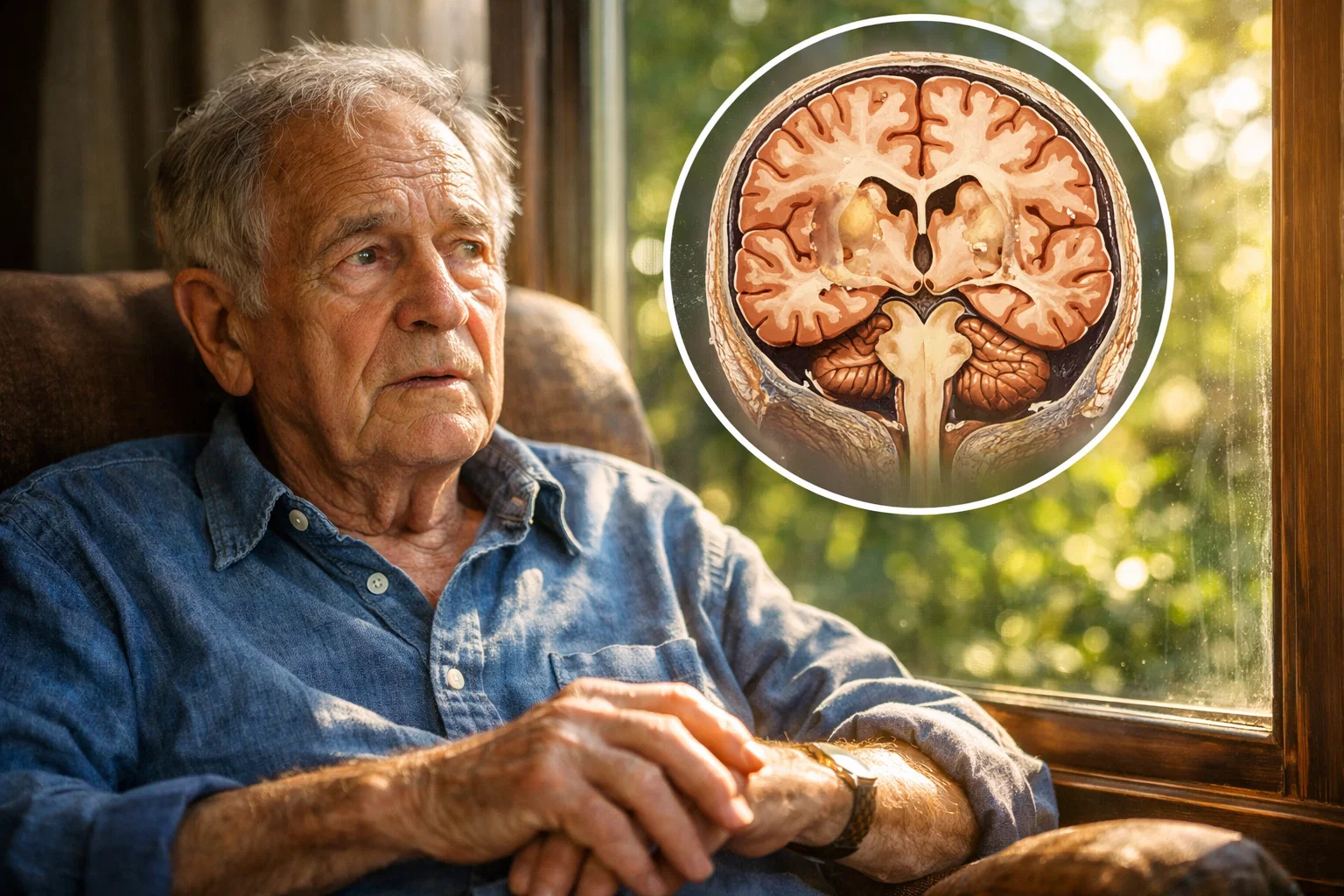
Dementia is still regarded as a fate of old age, as something that simply happens at some point, inevitably, genetically determined, and without being able to influence it. This view is convenient, but falls far short. It completely ignores what research and clinical experience have been showing for years: in most cases, dementia is not a sudden event, but the result of a long-term, often decades-long imbalance in the body.
Anyone who takes a holistic view of dementia recognizes it not only as a disease of the brain, but as a systemic process in which inflammation, metabolism, gut, hormones, nutrient supply, stress, and lifestyle interact. This is precisely where a great opportunity lies, because processes that develop slowly can often still be influenced in their dynamics or at least changed in their speed.
Dementia is not an isolated brain problem
The brain accounts for only about two percent of our body weight, yet it consumes about twenty percent of the total energy. It relies on a continuous supply of oxygen, glucose, fats, micronutrients, and hormonal signals. If this finely tuned system becomes unbalanced, mental performance suffers first, often long before a formal diagnosis is made.
It is important to distinguish normal fluctuations from pathological changes. Many people experience memory lapses, concentration problems, or word-finding phenomena without this having anything to do with neurodegenerative processes. Multilingualism, high mental demands, lack of sleep, stress, or hormonal shifts can temporarily slow down access to stored information without structures being lost. The brain does not work worse in these moments, but under different internal conditions.
In addition, mental performance is not constant, but follows an inner rhythm. Attention, language retrieval, working memory, and problem-solving ability fluctuate significantly throughout the day, and these fluctuations are genetically determined. Early, normal, and late types have very different time windows in which their prefrontal cortex, i.e., the brain region responsible for planning, language, and complex thinking, works optimally. A chronobiological late type may appear mentally significantly restricted in the early hours and show exceptional clarity and mental agility in the evening or at night.
This effect becomes particularly visible in people who use several languages. The brain manages several parallel language systems that constantly compete in everyday life. In biological low phases, this access becomes slower, words are “on the tip of the tongue” but first appear in another language or remain briefly blocked. In their own high phases, on the other hand, the same person functions effortlessly, precisely, and often even particularly creatively. These phenomena are an expression of a rhythmic, highly adaptive nervous system, not a beginning mental decline.
Changes only become relevant when they occur newly, intensify, noticeably impair everyday life, or persist regardless of time of day, sleep, and stress. Only in this context can they be indications of a system that is increasingly under biological overload.
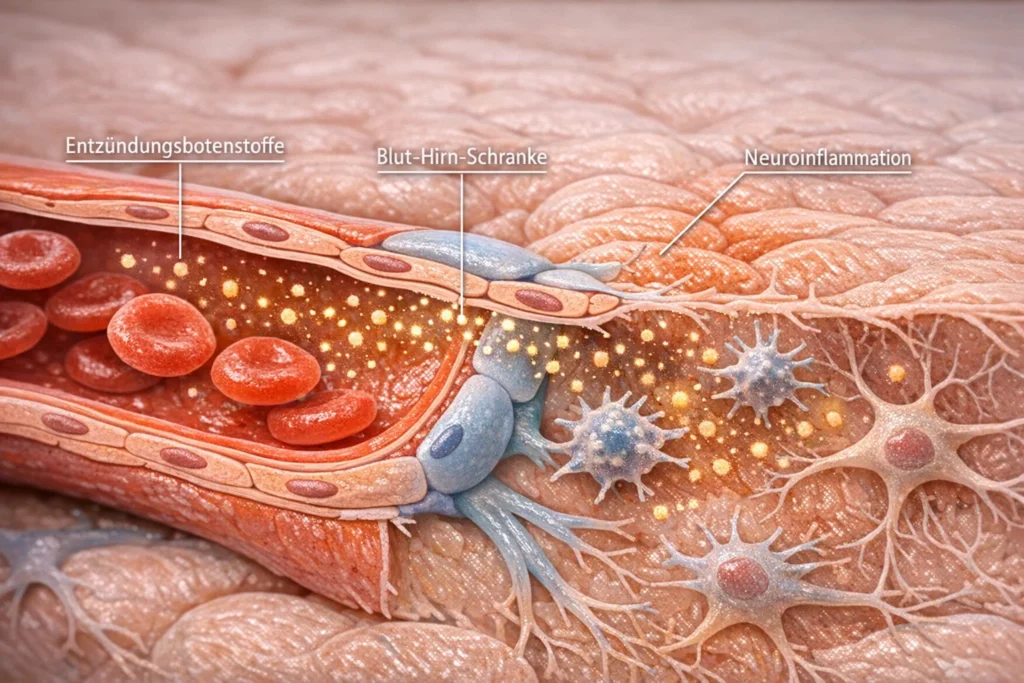
Chronic inflammation, the silent fire in the background
A central driver of this process is chronic, low-grade inflammation. Unlike acute inflammation, which is associated with pain, fever, or swelling, it develops quietly in the background. It arises when the immune system has to process small stimuli again and again over a long period of time, such as through blood sugar fluctuations, oxidative stress, bacterial fragments from the gut, lack of sleep, or permanent activation of the stress axes. These stimuli are not dramatic in themselves, but in their sum they keep the immune system in a state of constant alert.
Highly processed food, high sugar load, lack of exercise, chronic stress, and a disturbed intestinal flora amplify this subliminal activation. Immune cells begin to permanently release inflammatory messengers without a clear enemy being present. This creates an inflammatory environment that spreads insidiously throughout the body.
These inflammatory processes are not limited to individual tissues. The messengers also reach the brain via the bloodstream. They can make the blood-brain barrier more permeable and activate microglial cells there, which in turn trigger neuroinflammatory processes. In people with Alzheimer’s, such changes can often be detected long before the onset of clear symptoms.
Insulin resistance in the brain, the metabolic aspect of dementia
Parallel to the inflammatory processes, metabolism plays a central role. More and more research suggests that Alzheimer’s can also be understood as a form of insulin resistance in the brain. Nerve cells are dependent on glucose as their main source of energy, but this energy is only available to them if insulin enables its absorption into the cells. If this signaling effect is disturbed, a deficiency state arises, even though there is enough sugar in the blood.
A permanently unstable blood sugar with recurring insulin spikes leads to the insulin receptors becoming less sensitive over time. This mechanism, which contributes to classic insulin resistance in the rest of the body, also takes place in the brain. At the same time, inflammatory processes additionally promote this disorder, as inflammatory messengers directly interfere with the insulin signaling chains.
The consequence is that nerve cells can no longer use their energy efficiently. The brain enters a state of chronic energy deficiency, which initially manifests itself in concentration difficulties, mental fatigue, and reduced mental flexibility, and later in structural degradation. In this context, the term diabetes type 3 has emerged to describe the close connection between metabolic disorders and neurodegenerative processes.
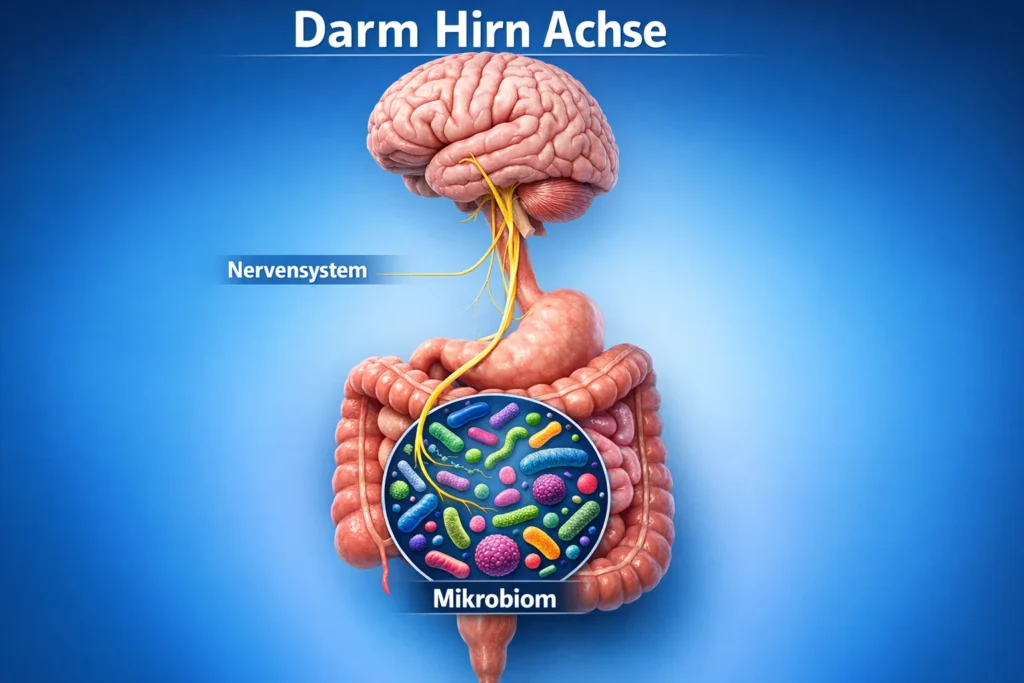
Gut and microbiome, the forgotten axis
The gut forms another, often underestimated control center in this process. Via the gut-brain axis, it is in constant exchange with the nervous system, the immune system, and the metabolism. Billions of microorganisms continuously produce messengers, metabolic products, and neurotransmitter precursors there, which act directly on the brain via nerve pathways, blood, and immune pathways.
If the microbiome has become unbalanced, for example due to diet, antibiotics, chronic stress, or inflammation, this signaling cascade changes. Certain bacteria produce fewer anti-inflammatory metabolites, while others increasingly release substances that activate the immune system. At the same time, the intestinal mucosa can become more permeable, so that bacterial components enter the bloodstream and amplify systemic inflammatory reactions.
These immunological signals also reach the brain. They can influence the blood-brain barrier, activate microglial cells, and amplify neuroinflammatory processes. At the same time, a disturbed intestinal function impairs the absorption and activation of many nutrients that are necessary for neuronal energy production, neurotransmitter formation, and antioxidant protection. This creates a connection between gut, inflammation, metabolism, and mental stability that goes far beyond digestion.
Nutrients as a prerequisite for mental stability
Many people with mental decline carry an invisible baggage of micronutrient deficiencies with them for years. The cause is rarely the amount of food, but its quality and the body’s ability to actually use what is ingested. An increasingly industrially influenced diet, combined with a weakened absorption due to stress, inflammation, and gut dysbiosis, insidiously shifts the internal environment into a state of chronic undersupply. The brain, one of the most nutrient-sensitive organs of all, reacts to this shift particularly early.
B vitamins and folic acid play a central role because they are involved in almost all processes that keep nerve cells functional. They control the formation of neurotransmitters, the structure of the myelin sheaths, the methylation of numerous genes, and the breakdown of homocysteine, a substance whose chronically elevated levels are associated with a significantly higher risk of mental decline. If these vitamins are lacking for a longer period of time, neuronal repair mechanisms, signal transmission, and vascular health come under increasing pressure.
Omega-3 fatty acids, especially DHA, form a structural component of the cell membranes in the brain. They influence the mobility of the membranes, the efficiency of signal transmission between nerve cells, and the readiness of the tissue for inflammation. If their availability decreases, the physical and biochemical stability of the nerve cells changes, making them more susceptible to inflammation, oxidative stress, and degenerative processes.
Magnesium acts as a buffer in the nervous system. It regulates the excitability of nerve cells, stabilizes synaptic functions, and protects against excessive activation. Chronic stress, increased cortisol levels, and a permanently high neuronal tension significantly increase magnesium consumption, which can lead to a silent deficiency that remains unnoticed for a long time, but makes the brain increasingly vulnerable.
Vitamin D acts as a regulatory hormone in the brain. It influences which genes are active in the brain and which are not, including many that control inflammation, signal transmission between nerve cells, and the adaptability of the brain. If the levels are too low, the brain becomes more susceptible to inflammation and loses part of its own protective function, which can have a long-term impact on mental stability. In addition, the blood levels considered sufficient in many European countries are comparatively low. While in Germany, levels around 20 ng/ml are often considered acceptable, functional target ranges in other countries and in preventive medicine are more in the range of 40 to 60 ng/ml. This creates a broad gray area in which people are formally considered to be supplied, but biologically already live in an undersupply that can have a long-term impact on the brain, immune system, and inflammation regulation. This prioritization is particularly impressive during breastfeeding, because the vitamin D content of breast milk depends directly on the vitamin D status of the mother. Only when the stores are well filled does the release to the infant increase significantly, an indication that the body initially retains vitamin D for its own stability and only passes it on generously when there is sufficient supply.
Zinc is closely linked to memory processes, synaptic communication, and the immune balance in the nervous system. Both deficits and oversupplies can impair neuronal function, which shows how finely balanced these trace elements act in the brain.
Iron also moves in a narrow biological corridor. A deficiency reduces the oxygen supply to the brain and thus the mitochondrial energy production, an excess promotes oxidative damage and inflammatory reactions. Both can undermine mental performance in the long term.
In summary, it shows that micronutrients are not isolated adjusting screws, but part of a highly sensitive system in which energy production, neurotransmission, immune regulation, and structural integrity interlock. If this system is not properly supplied for a longer period of time and loses its inner balance, the brain’s resistance, which actually protects it from degradation, also decreases.
For readers who would like to deepen this connection further, for example with regard to sugar, gluten, and alcohol as creeping disruptive factors of gut, inflammation, and nutrient utilization, my article “A little of it doesn’t hurt” offers a suitable extension of this thought.
Mitochondria and the creeping energy loss
At the cellular level, the mitochondria play a crucial role. They are the power plants of the cells and supply nerve cells in particular with the energy they need for signal transmission, repair processes, and plasticity. The brain is one of the most energy-hungry organs of all, which is why mitochondrial disorders become noticeable here particularly early.
Oxidative stress, chronic inflammation, nutrient deficiencies, and environmental toxins attack these sensitive structures over years. Free radicals, which are increasingly produced under stress, through inflammation, or through pollutants, damage the membranes and the DNA of the mitochondria. At the same time, in the case of poor nutrition or disturbed intestinal absorption, those vitamins, minerals, and fatty acids that would be necessary for their function are lacking. Permanently elevated blood sugar and insulin levels also burden the mitochondria because they make energy processing inefficient and trigger additional oxidative processes.
The consequence is a creeping loss of cellular energy. Nerve cells increasingly enter a state of deficiency in which they can no longer maintain their complex tasks. This energy deficiency initially manifests itself as mental fatigue, as declining concentration and lower resilience, and over time turns into structural degradation.
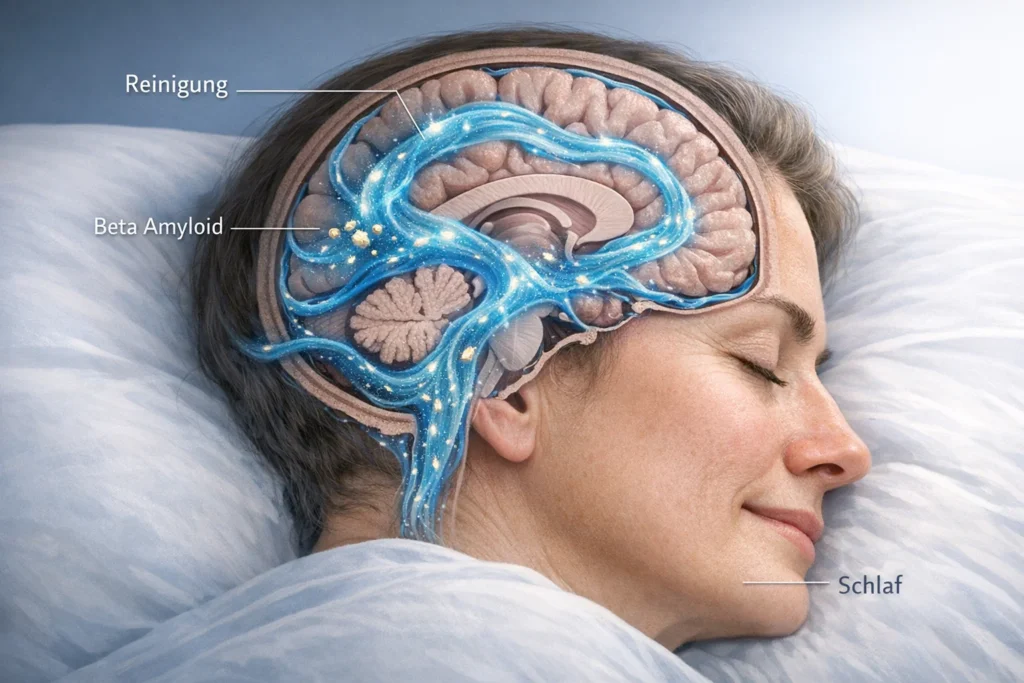
Sleep, stress, cortisol, and neuronal exhaustion
Chronic stress deeply interferes with the control of the brain. If the body remains in alarm mode for a long time, more cortisol is released, a hormone that is actually supposed to help deal with danger in the short term. However, if this state persists, it begins to burden exactly those structures that are important for learning, memory, and emotional stability. In the hippocampus, a central area for memory and orientation, the new formation of nerve cells is inhibited, while inflammatory processes increase and sugar metabolism becomes unbalanced. The brain remains trapped in a kind of permanent tension in which protective reactions dominate and regenerative processes become increasingly difficult to take hold.
Many people experience this state as inner restlessness, irritability, racing thoughts, or the feeling of never really coming to rest. The body is constantly trying to adapt, but without sufficient recovery, this adaptation becomes exhaustion. Particularly stressful is that this biological stress is often perceived in our culture as individual failure, as a lack of resilience or a lack of self-discipline, although it is actually a measurable shift in the nervous system.
Sleep is one of the most important counterpoles in this context. In the deep sleep phases, the brain shuts down its alarm programs and activates its repair mechanisms. Nerve cells are stabilized, connections are rearranged, metabolic waste is removed, and stress hormones are regulated. Just a few nights with bad or interrupted sleep are enough to noticeably impair concentration, memory, and emotional stability. If this state persists for months or years, the effect intensifies and contributes to the brain losing its resistance to inflammation, metabolic stress, and aging processes.
Why dementia is seen so late in society
On a social level, dementia also triggers fear. It touches on questions such as control, dependency, or also the question of identity. This fear often leads fatally to an inner turning away both in those affected, as well as in the environment and in institutional structures.
Early signs of forgetfulness, disorientation, or emotional changes are therefore often relativized or explained as signs of aging, as stress, or as a temporary phase. Behind this often lies the desire not to have to think about a possible diagnosis. In a culture that highly values functioning and autonomy, mental decline is experienced as a threat.
In addition, the health system is primarily geared towards acute events. Dementia, however, begins quietly over long periods of time. Between inconspicuous and clinically clear lies a long phase in which biological processes are already tipping without being clearly named. In this time, much would still be possible, but it is often overlooked.
Early attention would have a different quality here. It would not stigmatize, but make resources visible, enable relief, and stabilize the inner system as long as it is still flexible. The sooner we look, the greater the scope for action remains to help the body regulate itself again!
What early counter-regulation means
If dementia arises from a long biological imbalance, then this slowness also has its special meaning. The body does not lose its mental stability from one moment to the next, but in small shifts over years. And it is precisely in these shifts that direction can be given.
Early counter-regulation means changing the inner environment in which the brain ages. It is about the conditions under which nerve cells are permanently in survival mode or find access to repair, plasticity, and regeneration again.
This starts with the two basic biological rhythms of sleep and nutrition.
During sleep, the brain regenerates, breaks down waste products, stabilizes neuronal networks, and regulates stress hormones. During deep sleep phases, the glymphatic system becomes active, a cleaning system of the brain that removes metabolic waste products such as beta-amyloid, a protein that accumulates in harmful deposits in Alzheimer’s disease. If this process is disrupted over years due to lack of sleep, shift work, or inner restlessness, these substances remain in the tissue and promote neurodegenerative changes.
Nutrition also affects these processes because it continuously sends biochemical signals to the body. Every meal influences inflammation, blood sugar, insulin response, and the microbiome. A diet that is consistently high in sugar, heavily processed fats, and nutrient-poor products shifts this environment towards inflammation and metabolic instability. A nutrient-dense, plant-based, fiber-rich, and protein-rich diet without any refined sugar can shift this balance back in a different direction.
The stability of blood sugar is particularly crucial for the brain. It relies on a consistent supply of energy. Constant fluctuations create stress in the nervous system, amplify inflammatory processes, and, in the long term, promote insulin resistance, including in the brain. Meals that combine complex carbohydrates, plant-based fats, proteins, and fiber have a balancing effect. A plant-based diet, preferably of organic origin, offers all of this, as pesticides also burden the body, and especially the gut, as environmental toxins.
Through the gut, nutrition additionally affects neuroinflammation, i.e., inflammatory processes in the brain. Fiber, polyphenols, and fermentable plant substances, all of which are abundant in a plant-based diet, nourish the very bacteria in the gut that form short-chain fatty acids and stabilize the gut-brain axis. In this way, the susceptibility to inflammation in the brain can be positively influenced long before structural damage occurs.
The supply of micronutrients is also part of this early counter-regulation. B vitamins, omega-3 fatty acids, magnesium, vitamin D, zinc, and a balanced iron supply form the basis for nerve cells to remain stable, for the signaling substances in the brain to be in balance, and for the mitochondria, i.e., the power plants of the cells, to be able to provide sufficient energy. Deficiencies develop slowly and often go unnoticed for a long time, but over the years, they can weaken the brain’s resilience.
Stress regulation
In addition to sleep and nutrition, the nervous system decisively shapes the internal climate in which the brain lives. If the body remains in a state of alert for a long time because stress, overwhelm, or inner insecurity do not subside, the entire system is geared towards survival rather than regeneration. Inflammatory processes increase, blood circulation changes, sleep becomes shallower, and the ability to form new neuronal connections decreases. The brain then no longer works from a state of stability but from permanent adaptation.
Rhythm, breathing, movement, social security, and conscious breaks act as biological counterweights in this context. They signal to the nervous system that there is no acute danger and allow it to switch from alert mode to a state in which repair, learning, and regeneration can take hold again. These seemingly simple factors determine whether inflammation is dampened or further fueled, whether energy is built up or consumed.
All these levels interlock. They change not only individual values but the entire environment in which the brain ages. This is precisely where the actual prevention lies, in the conscious shaping of those conditions under which mental stability can arise and be maintained in the first place.
Dementia is not a sudden fate
Dementia is rarely a sudden fate. It is usually the result of a long history of biological overload, in which inflammation, metabolism, stress, sleep deprivation, nutrient deficiencies, and social stresses act on the same system for years. Anyone who considers it holistically recognizes not only the risks but also those areas in which influence remains possible, long before a diagnosis seems inevitable.
Not everything can be prevented. Genes, life history, environmental burdens, and age shape a person’s path, but at the same time, much can be influenced or slowed down if the brain, body, and life context are understood as an interconnected system. A brain does not age on its own! It ages in a body, in a nervous system, in everyday life, in relationships, in rhythms, in eating habits, and in an inner and outer environment as a whole.
This is also where epigenetics comes in. It describes those biological mechanisms through which environment, nutrition, stress, sleep, and emotional experiences control which genes are active in a cell and which remain silent. The genes themselves do not change, but their behavior, i.e., what information they pass on, continuously reacts to the internal and external environment. In this way, it becomes understandable why lifestyle, burdens, and caregiving deeply intervene in the biological processes that, in the long term, help determine health or disease.
Holistic health means taking these connections seriously and supporting people in regaining influence over their internal environment. Understanding and perception, as well as the conscious shaping of what can be shaped, form the basis for this.
Thus, the view of dementia becomes a view of a process that, like all biological processes, is related to the conditions under which it arises.
Do you find my posts informative and don’t want to miss any? Then join my subscribers and stay up to date on everything you need to know via the newsletter.
Sources:
- Sleep, glymphatic system, removal of waste products such as beta-amyloid
Xie L. et al. (2013). Sleep drives metabolite clearance from the adult brain. Science.
https://www.science.org/doi/10.1126/science.1241224
Summary: Animal experimental data show that the interstitial space increases during sleep and the removal of metabolites, including beta-amyloid, is significantly more efficient than in the waking state. - Sleep deprivation and beta-amyloid in humans
Shokri-Kojori E. et al. (2018). β-Amyloid accumulation in the human brain after one night of sleep deprivation. PNAS.
https://www.pnas.org/doi/10.1073/pnas.1721694115
Summary: PET data suggest that one night of sleep deprivation may be associated with measurably increased beta-amyloid load in certain brain regions. - Lifestyle works if you start early, multidomain prevention (nutrition, exercise, cognitive training, vascular risks)
Ngandu T. et al. (2015). A 2 year multidomain intervention… (FINGER trial). The Lancet.
https://pubmed.ncbi.nlm.nih.gov/25771249/
Summary: Randomized study in at-risk individuals, a structured combination of lifestyle measures was able to improve or stabilize cognitive functions compared to the control. - Insulin resistance in the brain, “Type 3 diabetes” as a concept
de la Monte S.M. (2008). Alzheimer’s disease is type 3 diabetes, evidence reviewed. J Diabetes Sci Technol.
https://pmc.ncbi.nlm.nih.gov/articles/PMC2769828/
Summary: Review summarizing that disturbances in insulin, IGF signaling pathways in the brain may be early and progressive features in Alzheimer’s, including mechanistic animal models and overlaps with metabolic pathologies. - Inflammation and Alzheimer’s, neuroinflammation as a central process
Heneka M.T. et al. (2015). Neuroinflammation in Alzheimer’s disease. The Lancet Neurology.
https://pubmed.ncbi.nlm.nih.gov/25792098/
Summary: Overview of microglia, astroglia, and innate immune responses in the brain, with the key point that inflammatory mechanisms play a relevant role in pathogenesis and progression. - Homocysteine as a risk factor, B vitamins as an approach to modulation
Smith A.D. et al. (2018). Homocysteine and Dementia, An International Consensus Statement.
https://pubmed.ncbi.nlm.nih.gov/29480200/
Summary: Consensus overview describing homocysteine as a modifiable risk factor and discussing interventions with B vitamins in the context of atrophy rate and cognitive course, with clear classification of the evidence. - Homocysteine, dose-response relationship, and dementia risk
Zhou F. et al. (2019). Dose-response meta-analysis… blood homocysteine and Alzheimer-type dementia risk.
https://pubmed.ncbi.nlm.nih.gov/30826501/
Summary: Meta-analysis of prospective data, higher homocysteine levels are dose-dependently associated with a higher risk of Alzheimer’s dementia. - Dietary patterns, MIND diet, and Alzheimer’s incidence
Morris M.C. et al. (2015). MIND diet associated with reduced incidence of Alzheimer’s disease.
https://pubmed.ncbi.nlm.nih.gov/25681666/
Summary: Cohort-based associations, higher adherence to the MIND diet correlates with lower Alzheimer’s incidence, even moderate adherence showed effects. - MIND diet and dementia risk in a larger population
Chen H. et al. (2023). Association of the MIND Diet With the Risk of Dementia. JAMA Psychiatry.
https://jamanetwork.com/journals/jamapsychiatry/fullarticle/2804268
Summary: Observational data, MIND adherence is associated with a lower risk of incident dementia, with the usual caveat that these are associations, not causal evidence. - Vitamin D and dementia risk, meta-analysis
Kalra A. et al. (2020). Association of Vitamin D Levels with incident all-cause dementia and Alzheimer’s disease, a meta-analysis.
https://www.sciencedirect.com/science/article/pii/S2274580724004618
Summary: Meta-analysis of prospective studies, low vitamin D levels are associated with a higher risk of dementia and Alzheimer’s, effect sizes vary depending on cut-offs and study design. - Microbiome and Alzheimer’s spectrum, systematic review and meta-analysis
Hung C.C. et al. (2022). Gut microbiota in patients with Alzheimer’s disease spectrum, systematic review and meta-analysis.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8791218/
Summary: Summary of studies reporting recurring differences in composition and diversity of the gut microbiota in the Alzheimer’s spectrum compared to controls, including classification of the limitations. - Exercise as a modifiable risk factor, meta-analysis of prospective cohorts
Zhang X. et al. (2023). Effect of physical activity on risk of Alzheimer’s disease, a meta-analysis of prospective cohort studies.
https://pubmed.ncbi.nlm.nih.gov/37979700/
Summary: Higher physical activity is consistently associated with a lower Alzheimer’s risk in prospective cohorts, despite inevitable confounding issues in observational data.